 The project workplan will be driven by three main methodological lines:
The project workplan will be driven by three main methodological lines:
1. The Clinicians, the main target of OraMod, will be constantly at the centre of the development process.
2. Exploitation related issues will be addressed from the beginning: market and technology standards, product certification, market demand, technology progress and competition scenarios.
3. The Consortium will keep a constant link with the VPH Community: the opensource toolkit published by VPH NoE will also be considered for use in the development phase.
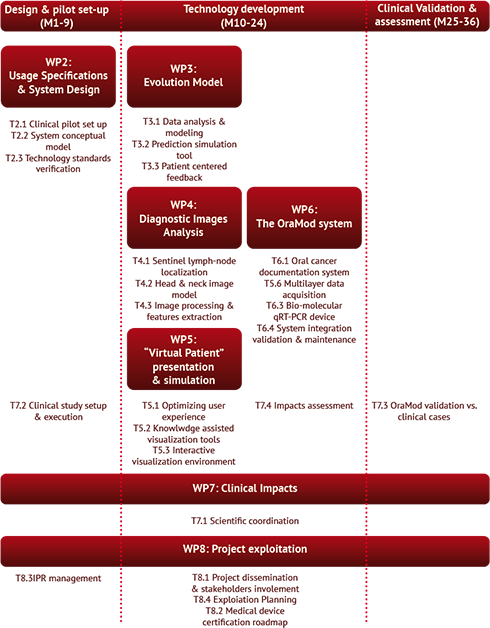
The project work will be organised into three main steps: (1) the design and pilot set-up, concentrated in the first 9 months, (2) the technical development up to the end of the second year, and (3) the clinical validation and assessment in the last year of the project. OraMod R&D spans from IT to advanced biology and specialized oncology areas, and involves all levels of complexity in the area of human physiology as well as of modelling techniques and system engineering, therefore a strict and effective management procedure has been envisaged, both at scientific and technical level.
The involvement of 4 reference European hospitals for Oral Cavity cancer will provide a large number of cases for the optimization and validation of the OraMod model and for the assessment of the relevant clinical and socio-economic impacts.